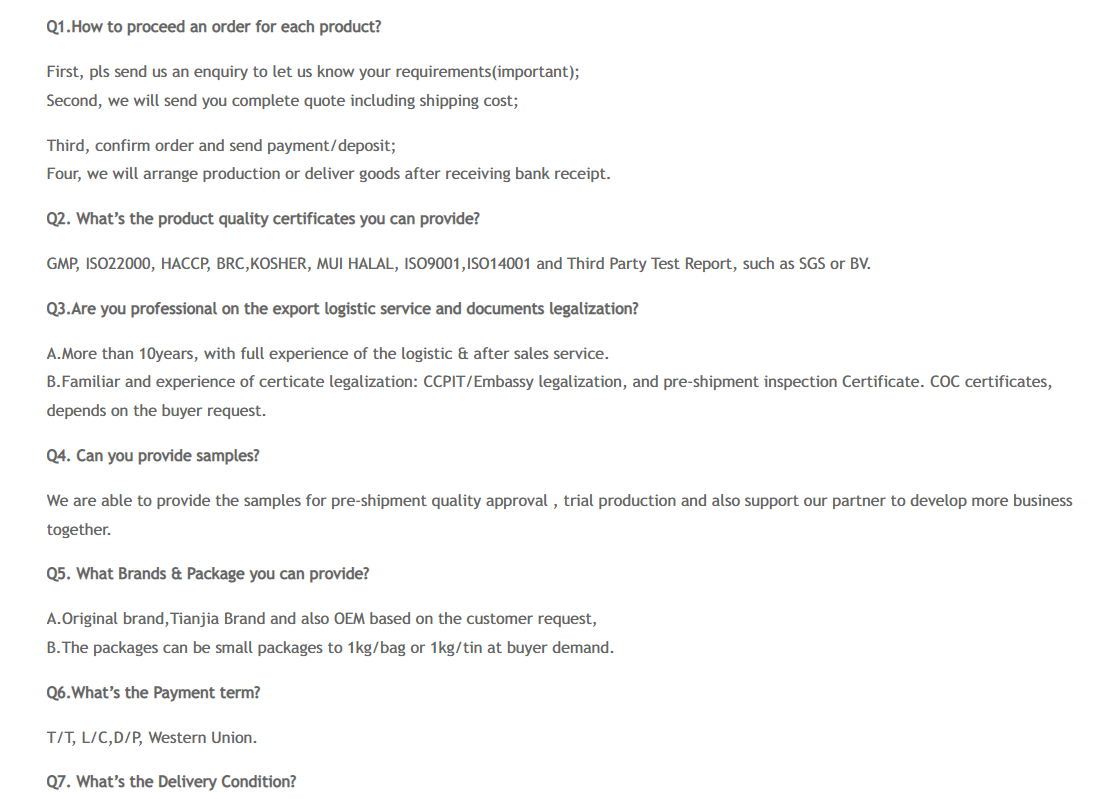
Manufacturer Good Price CALCIUM CHLORIDE CAS: 10043-52-4
Applications of CALCIUM CHLORIDE
1. Calcium chloride (CaCl2) has many uses. It is used as a drying agent and to melt ice and snow on highways, to control dust, to thaw building materials (sand, gravel, concrete, and so on). It is also used in various food and pharmaceutical industries and as a fungicide.
2. Calcium chloride is one of the most versatile of the basic chemicals.It has several common applications such as brine for refrigeration plants, ice and dust control on roads, and in concrete. The anhydrous salt is also widely used as a desiccant, where it will absorb so much water that it will eventually dissolve in its own crystal lattice water (water of hydration). It can be produced directly from limestone, but large amounts are also produced as a by-product of the “Solvay Process” (which is a process to produce soda ash from brine).
Calcium chloride is also commonly used as an additive in swimming pool water as it increases the “calcium hardness” value for the water.Other industrial applications include use as an additive in plastics, as a drainage aid for wastewater treatment, as an additive in fire extinguishers, as an additive in control scaffolding in blast furnaces, and as a thinner in “fabric softeners”.
Calcium chloride is commonly used as an “electrolyte” and has an extremely salty taste, as found in sports drinks and other beverages such as Nestle bottled water. It can also be used as a preservative to maintain firmness in canned vegetables or in higher concentrations in pickles to give a salty taste while not increasing the food’s sodium content. It is even found in snack foods, including Cadbury chocolate bars.In brewing beer, calcium chloride is sometimes used to correct mineral deficiencies in the brewing water. It affects flavor and chemical reactions during the brewing process, and it can also affect yeast function during fermentation.
Calcium chloride can be injected as intravenous therapy for the treatment of “hypocalcemia” (low serum calcium). It can be used for insect bites or stings (such as Black Widow spider bites), sensitivity reactions, particularly when characterized by “urticaria” (hives).
3. Calcium Chloride is a general purpose food additive, the anhydrous form being readily soluble in water with a solubility of 59 g in 100 ml of water at 0°c. it dissolves with the liberation of heat. it also exists as calcium chloride dihydrate, being very soluble in water with a solubility of 97 g in 100 ml at 0°c. it is used as a firming agent for canned tomatoes, potatoes, and apple slices. in evaporated milk, it is used at levels not more than 0.1% to adjust the salt balance so as to prevent coagulation of milk during sterilization. it is used with disodium edta to protect the flavor in pickles and as a source of calcium ions for reaction with alginates to form gels.
4. Obtained as a by-product in the manufacture of potassium chlorate. The white crystals, soluble in water and alcohol, are deliquescent and must be kept in a well-stoppered bottle. Calcium chloride was used in iodized collodion formulas and in collodion emulsions. It was also an important desiccating substance used in tin calcium tubes designed to store presensitized platinum papers.
5. For the treatment of hypocalcemia in those conditions requiring a prompt increase in blood plasma calcium levels, for the treatment of magnesium intoxication due to overdosage of magnesium sulfate, and used to combat the deleterious effects of hyperkalemi
6. Calcium chloride is highly hygroscopic and is often used as a desiccant.
7. Calcium chloride is an astringent. It also helps improve the reaction among certain ingredients used in cosmetic formulations. This inorganic salt is no longer commonly used in skin care products and is being replaced with potassium chloride.
Specification of CALCIUM CHLORIDE
|
Compound |
Specification |
|
APPEARANCE |
WHITE,HARD ODORLESS FLAKE, POWDER,PELLET,GRANULE |
|
CALCIUM CHLORIDE(As CaCl2) |
94% min |
|
MAGNESIUM&ALKALI METAL SALT (As NaCl) |
3.5% max |
|
WATER INSOLUBLE MATTER |
0.2% max |
|
ALKALINITY(As Ca(OH)2) |
0.20% max |
|
SULFATE (As CaSO4) |
0.20% max |
|
PH VALUE |
7-11 |
|
As |
5 ppm max |
|
Pb |
10 ppm max |
|
Fe |
10 ppm max |
Packing of CALCIUM CHLORIDE
25KG/BAG
Storage: Calcium chloride is chemically stable; however, it should be protected from moisture. Store in airtight containers in a cool, dry place.


Our Advantages

FAQ